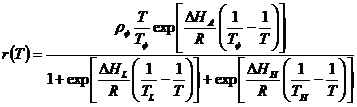|
Mealybug, Phenacoccus solenopsis Tinsley, is emerging as a key insect pest on Bt cotton since 2005. White waxy build-up on terminals, stems and branches of infested plants accompanied by intense activity of ants is a sure sign of mealybug attack. The bugs suck sap from plants leading to stunted growth and drying. Black moulds grow on the honeydew secretions of the mealy bug. Its natural spread by wind, water, ants, intercultural operations and other human activities like movement of infested material contributed to its quick colonization of new areas. High temperatures along with low humidity are congenial for its rapid growth and multiplication, while high intensity rains and wet spells adversely affect its infestation.
Introduction
Widespread infestation of the mealybug, Phenacoccus solenopsis Tinsley (Hemiptera:Pseudococcidae) on cotton in India occurred over large areas across the three cotton growing regions, attaining pest status in 2008. P. solenopsis has been the topic of research for insect taxonomists and applied entomologists in India due to its invasiveness, rapid spread, morphological and biological variations and the need for establishing an effective control strategy. Crops of different categories viz., food, fibre, fruit, ornamental, plantation and vegetable crops besides weeds are infested by P. solenopsis. P. solenopsis has a wider geographical distribution in its origin in Central America followed by reports of the Caribbean and Ecuador, Chile, Argentina, Brazil. P. solenopsis has been described as a serious cum invasive pest of cotton in Pakistan and India, and on Hibiscus rosasinensis in Nigeria. P. solenopsis has been reported in China on cotton and hibiscus. Widespread infestation of P. solenopsis and economic damage to cotton across nine cotton growing States of the country viz., Punjab, Haryana, Rajasthan, Gujarat, Madhya Pradesh, Maharashtra, Andhra Pradesh, Karnataka and Tamil Nadu during 2008-09 crop season necessitated a national level consultation at central Institute for Cotton Research (CICR), Nagpur towards formulation of strategies for its management[1-4]. Survey across 47 locations of the country between months of late 2007 and early 2008 established the predominance of P. solenopsis [5] in India. The solenopsis mealybug exhibits obligate sexual ovoviviparous reproduction. Female mealybug Female mealybug life cycle consists of five stages (egg, 1st, 2nd and 3rd instar nymph and adult) [13]. The adult is wingless with a 3-4 mm long oval shaped body which is covered with white hydrophobic (water repellent) mealy wax. There are dark bare spots on the thorax and abdomen, which appear as dark longitudinal lines. P. solenopsis was observed to be ovoviviparous. Mature females lay eggs in waxy pouches called ovisacs. Both eggs and crawlers were observed in the ovisac and the eggs hatched within a short period (within 1 d) after deposition. Male mealybug The adult male mealybug life cycle consists of six stages (egg, 1st and 2nd instar nymph, pre-pupa, pupa and adult). The adult is about 1 mm long, with a grey body and a single pair of transparent wings. Two filaments of white wax project from the end of its abdomen. The adult male has reduced mouthparts and causes no damage. The infestation based on presence or absence of mealy bugs and the severity using zero to four scale of infestation viz., 0 â No mealybug; 1 â Scattered appearance of few mealybugs on the plant; 2 â Severe incidence of mealybug on any one branch of the plant; 3 â Severe incidence of mealybug on more than one branch or half portion of the plant and 4 â Severe incidence of mealybug on the whole plant was recorded. Study on sample size indicated the importance of locating the source of mealybug infestation first, and sampling that accounts field areas largely parallel to the infestation source. While sample sizes of 25 to 50 plants per acre are sufficient in fields with known source of infestation such as roadside weeds and water channels, 100 plants per acre have to be sampled in clean field where prior knowledge of mealybug infestation is not available [6]. Mealybugs are small sap-sucking insects cause severe economic damage to cotton and a wide range of vegetable, horticultural and other field crops. Plants infested by mealybugs during vegetative phase exhibit symptoms of distorted, bushy shoots, crinkled and/or twisted bunchy leaves and stunted plants that dry completely in severe cases. Late season infestations during reproductive crop stage result in reduced plant vigour and early crop senescence. While feeding mealybugs inject a toxic substance into the plant parts resulting in chlorosis, stunting, deformation and death of plants [8]. At the Central zone, with the cotton + pigeonpea cropping system, the potential reduction in yield levels in respect of Grade 1, Grade 2, Grade 3 and Grade 4 severity of mealybug infestation was estimated to be 36.5, 46.6, 63.5 and 76.4% for M. hirsutus and 2.4, 31.5, 39.9 and 43.9 % for P. solenopsis indicating the potential of former over the later species. Since the appearance of Grade 1 symptom due to M. hirsutus occurred always with the presence of reproducing females along with crawlers on the plants, the yield losses had been higher over P. solenopsis. Nevertheless, fewer numbers of crawlers of P. solenopsis alone led to Grade 1 that caused insignificant yield loss in cotton. In the North zone, the reduction yield of cotton plants due to P. solenopsis was estimated to be 14.6 and 53.6 per cent at first and fourth grade mealybug infestation levels, respectively [7]. Development of symptoms among infested cotton plants occur much faster with M. hirsutus over P. solenopsis. While Grade 2 severity of M. hirsutus could cause severe stunting of 30-40 days old cotton crop, only extreme severity (Grade 4) of P. solenopsis affected the crop growth symptomatically. In addition, the highly aggregative and intense within plant attribution of M. hirsutus at severity levels of Grade 3 and 4 had caused higher yield losses over P. solenopsis. However, the diverse and higher levels of parasitisation of M. hirsutus in Central cotton production system had kept its occurrence sporadic and limited [7]. A total of 194 host plans belonging to cultivated crop plant species, ornamentals, and weed species were recorded to support P. solenopsis during the season and off-season. The diversity of hosts for P. solenopsis was greater in Central (72%) followed by South (64%) and North (37%) zones. Out of the total 194 hosts of P. solenopsis documented across the country, 55.6% were weeds. Highest number of host plants belonged to Solanaceae (10), Asteraceae (17), Fabaceae (14) and Malvaceae (23) [7, 11 & 12]. The effect of temperature on life cycle of the solenopsis mealybug, Phenacoccus solenopsis Tinsley, on cotton (Gossypium hirsutum L.) was assessed under laboratory conditions at ten constant temperatures (18â40 A°C)[9]. The development duration of female and male nymphal instars linearly decreased with the increase in temperature from 18 to 32 A°C. Cumulative developmental time of females ranged from 43.9 d (18 A°C) to 15.0 d (32 A°C). Survival of crawlers to adulthood was lowest (<53%) at 20 and 36 A°C and highest (80%) at 32 A°C. The solenopsis mealybug exhibited obligate sexual ovoviviparous reproduction and the pre-oviposition period in mated females showed a significant decreasing trend between 20 A°C (23.0 d) and 30 A°C (9.5 d). The oviposition period of 10.2â11.5 d at â¥25 A°C was nearly half the duration than at 20 A°C and the highest fecundity (245 eggs + crawlers) was observed at 30 A°C. Longevity of mated females was significantly prolonged at 20 A°C (46.0 d) compared to 30 A°C (21.4 d). Proportion of females was highest (97.5%) at 25 A°C. Males required higher degree-days (363.6) for their cumulative development compared to females (317.5). Lower temperature thresholds estimated from the linear model for cumulative female and male development were 11.7 and 10.1 A°C, respectively. The estimated optimum temperature thresholds for nymphal instars (32â33.4 A°C) from β type distribution function were closer to the observed maximum developmental rate compared to Lactin-2 model. The population trend index using survival, fecundity, and sex ratio of P. solenopsis with an initial population of 100 crawlers in the Morris-Watt life table model indicated a potential population increase of 170.3 and 97.6 times at 30 and 35 A°C, respectively, in the next generation. Temperature-driven development and survival rates of the mealybug, Phenacoccus solenopsis Tinsley (Hemiptera: Pseudococcidae) were examined at nine constant temperatures (15, 20, 25, 27, 30, 32, 35 and 40A°C) on hibiscus [10]. Crawlers successfully completed development to adult stage between 15 and 35A°C, although their survival was affected at low temperatures. Two linear and four nonlinear models were fitted to describe developmental rates of P. solenopsis as a function of temperature, and for estimating thermal constants and bioclimatic thresholds (lower, optimum and upper temperature thresholds for development: Tmin, Topt and Tmax, respectively). Estimated thresholds between the two linear models were statistically similar. Ikemoto and Takai’s linear model permitted testing the equivalence of lower developmental thresholds for life stages of P. solenopsis reared on two hosts, hibiscus and cotton. Thermal constants required for completion of cumulative development of female and male nymphs and for the whole generation were significantly lower on hibiscus (222.2, 237.0, 308.6 degree-days, respectively) compared to cotton. Three nonlinear models performed better in describing the developmental rate for immature instars and cumulative life stages of female and male and for generation based on goodness-of-fit criteria. The simplified β type distribution function estimated Topt values closer to the observed maximum rates. Thermodynamic SSI model indicated no significant differences in the intrinsic optimum temperature estimates for different geographical populations of P. solenopsis. The estimated bioclimatic thresholds and the observed survival rates of P. solenopsis indicate the species to be high-temperature adaptive, and explained the field abundance of P. solenopsis on its host plants. Linear models Two models were evaluated to estimate the linear relationship between ecologically relevant temperatures and the rate of development of P. solenopsis. The first was the thermal summation model [14] which is given by the expression,
where, r is the rate of development (=1/ Development time (D) in days), T is ambient temperature (oC); intercept (a) and slope (b) are the model parameters. Thermal constant, k (= 1/b), is the number of degree-days (DDs) or heat units above the threshold needed for completion of an instar (Table S1). Lower temperature threshold (Tmin) was determined as the x-intercept (= - a/b) which is the estimated lower temperature at which the rate of development is either zero or no measurable development occurs. Standard error (SE) values of Tmin and k were calculated as described by Campbell [14]. The second linear model by Ikemoto and Takai [15] is given as,
where, DT is the product of the duration of development, D (days), and temperature, T (oC), k is thermal constant and Tmin is the lower developmental threshold. Nonlinear models Three empirical nonlinear models were fitted to the instar specific developmental rate data to estimate the optimum temperature threshold (Topt) and upper temperature threshold (Tmax). Topt is the threshold temperature at which developmental rate is maximal, while Tmax is the lethal threshold at which development ceases. Lactin-2 model [16], Briere-1 model [17] and simplified β type distribution function [18] were applied to assess the nonlinear relationship. In addition, a thermodynamic model referred to as the Sharpe-Schoolfield-Ikemoto model (SSI model) was used to estimate intrinsic optimum temperature for P. solenopsis [19]. All the nonlinear models described the relationship between developmental rate (1/D) and temperature (T). The Lactin-2 model is given by the expression,
where, D is the mean development duration in days, Ï is the composite value for critical enzyme-catalyzed biochemical reactions as T increases to Topt, â is the difference between Topt and Tmax when thermal breakdown becomes the overriding influence and l is a fitted coefficient that forces the nonlinear curve to intersect the x-axis and allows the estimation of lower developmental threshold. Although Tmax is a parameter in the Lactin-2 model, it does not actually represent the upper temperature at which growth rate equals to zero (the upper developmental threshold). The true developmental threshold predicted by the model can be obtained only by simulation. Thus both Tmin and Tmax were numerical approximations obtained as the roots of the fitted model by running the Newton-Raphson algorithm (SAS 9.2). Similarly, Topt was obtained from the Lactin-2 equation by iterating the temperature parameter until the developmental rate was maximized. The Briere-1 model is given by the expression,
where, r is the developmental rate as a function of temperature (T), and ‘a’ is an empirical constant. The simplified β type distribution function fitted to the data is given by the expression,
where k, α and β are model parameters estimated by Marquardt’s nonlinear method. In this model, Tmax and Topt could not be derived directly from the equation as parameters. Tmax was estimated graphically from the rapid decline of the right descending branch and Topt was derived using optimization. The nonlinear thermodynamic SSI model [20] is given by the expression:
where, developmental rate (r) is a function of temperature, T (in absolute temperature, K) (273.15K = 0A°C), R is the gas constant (1.987 cal/deg/mol), âHA is the enthalpy of activation of the reaction that is catalyzed by the enzyme (cal/mol), âHL is the change in enthalpy associated with low-temperature inactivation of the enzyme (cal/mol), âHH is the change in enthalpy associated with high-temperature inactivation of the enzyme (cal/mol), TLis the temperature at which the enzyme is half active and half low-temperature inactive (K), TH is the temperature at which the enzyme is half active and half high-temperature inactive (K), TÏ is the intrinsic optimum temperature at which the probability of enzyme being in the active state is maximal (K), and ÏÏ is the mean development rate at the intrinsic optimum temperature (TÏ) assuming no enzyme inactivation (day-1).
|